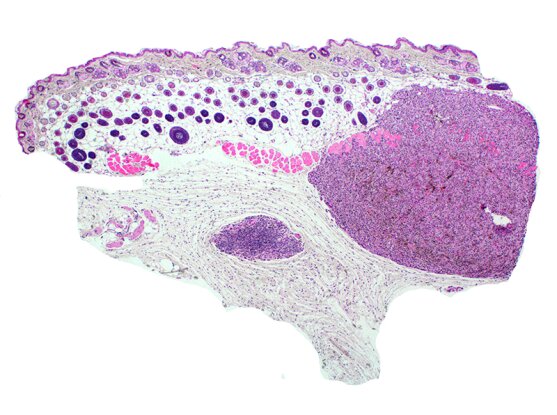
By introducing cancer-causing mutations into healthy skin cells step-by-step, Broad scientists have created models of skin cancer that can reveal the effects of mutations.
Over the last two decades, researchers have discovered thousands of genetic mutations in cancer, but understanding how they affect the growth and spread of tumors in the body remains challenging because each patient’s tumor can have many different mutations.
Now, scientists at the Broad Institute of MIT and Harvard have used the CRISPR-Cas9 gene-editing system to build cell models of melanoma, the deadliest skin cancer. In Science, the team describes how they installed five melanoma mutations, one-by-one and in different combinations, in the genome of healthy human skin cells. The edited cells grew and multiplied to become tumors that showed hallmarks of melanoma, including rapid growth, heightened ability to invade other tissues, the activation of certain gene programs, and specific pigmentation patterns.
Because the researchers introduced mutations one at a time in a controlled manner, they were able to pinpoint the effects of individual and specific combinations of mutations. They even found a causal link between one mutation and metastasis, the spread of tumors in the body. The results not only shed light on key mutations in melanoma, but also suggest a new way to study the role of specific genes in other cancers.
Eran Hodis, a study co-first and co-corresponding author, said the work is the first time scientists have made a human cancer model using precisely controlled genetic engineering and starting from fully differentiated, or specialized, cells rather than from stem cells, which are not relevant for every cancer type. Hodis began the study during his Ph.D. at Broad, the Dana-Farber Cancer Institute, and Harvard University, and is now an internal medicine resident at Brigham and Women’s Hospital. “It’s our hope that this approach will open up opportunities to build similar models across many other cancer types, accelerating the linking of cancer genetics to specific disease features,” he said.
“Having this toolbox enables us to ask what effects mutations have in cells,” said co-first author Elena Torlai Triglia, a postdoctoral scholar at the Broad. “These questions help us understand how the disease develops and how to target it.”
“To devise these models, we had to combine cutting-edge, precise gene editing technology with high-resolution, massively parallel single-cell genomics assays to generate and characterize the cells and tumors, along with machine learning algorithms to analyze the data. Together, these gave us an unprecedented view into what mutations and their combinations cause cancer in cells,” said Aviv Regev, co-corresponding and senior author of the study who was a core institute member at the Broad when the study began and is currently head of Genentech Research and Early Development.
Making melanoma
Connecting a tumor’s genetic makeup, or genotype, with a particular trait, or phenotype, can be particularly difficult when studying melanoma. “The skin is exposed to a lot of external agents, such as UV light, so there are a lot of mutations present in melanoma patients that might not be the ones really driving the disease,” Torlai Triglia said. By introducing mutations into healthy human melanocytes—skin cells that produce a pigment called melanin and become cancerous in melanoma—the team could observe the effects of the mutations on a blank slate.
To make their melanoma models, the team used Cas9, the CRISPR editing protein, to install mutations commonly seen in melanoma in the CDKN2A, BRAF, and TERT genes. Together, the three mutations caused the cells to behave like cancer, dividing indefinitely.
Next, the researchers added different combinations of additional mutations in known melanoma genes, PTEN, TP53, and APC, resulting in nine different cell models that the team then implanted in mice. The animals developed tumors that showed similar pigmentation and cellular architecture to human tumors. Single-cell RNA sequencing revealed that as more mutations were added, the cells gradually shifted their gene expression programs, and that the animal tumor models had similar patterns of gene expression as patient tumors with the same genotype—something the researchers were surprised to observe. “This would be such an impossible thing for us to mimic if we set out to do that in the lab,” Hodis said. “We’re talking about the coordinated expression of thousands of genes. This is one of the best pieces of evidence we have that the melanomas we built ‘from scratch’ faithfully reflect melanomas that arise in patients.”
Melanomas are notorious for metastasizing early—an often lethal process with an unclear genetic basis. Hodis, Torlai Triglia, and their collaborators found that metastasis often occurred in tumors with an APC mutation, suggesting that inactivation of the gene could contribute to the ability of tumors to spread.
Hodis, Torlai Triglia, and Regev plan to use their method to build more cancer models and study mutations that aren’t well understood. They said their findings underscore the importance of studying interactions between genes in models of disease, and that their approach may help scientists study how tumors become resistant to targeted therapies, which can’t always be explained by a single mutation.
Source: Read Full Article