Prediabetes is a condition that precedes type 2 diabetes and increases the risk of heart attack, kidney and eye disease, and several types of cancer. There is no approved drug therapy for prediabetes. Scientists at the German Center for Diabetes Research (DZD) now show how and by which mechanisms prediabetes can be brought into remission, i.e., into a state in which blood glucose levels return to normal.
The study shows that this protects against type 2 diabetes and is associated with better kidney and vascular function in the long term. The underlying mechanisms are different from those in type 2 diabetes remission, the researchers report in The Lancet Diabetes & Endocrinology.
People with type 2 diabetes have an increased risk of heart attack, kidney disease, and stroke, and a higher mortality risk. Type 2 diabetes was thought to be irreversible until a few years ago. We now know that type 2 diabetes can be put into remission in a significant number of individuals through substantial weight loss. However, this remission rarely lasts as most people typically develop type 2 diabetes again within a few years.
“We aimed to explore the feasibility of commencing earlier and implementing preventive measures already at a stage that precedes type 2 diabetes, namely prediabetes, with the aim of reversing it,” says senior author Prof. Dr. Andreas Birkenfeld, Medical Director of Medical Clinic IV at Tübingen University Hospital, and Director of the Institute for Diabetes Research and Metabolic Diseases of Helmholtz Munich at the University of Tübingen. This could be crucial for patients with prediabetes as they are at increased risk of developing type 2 diabetes as well as heart, kidney, and eye complications among others.
But what causes prediabetes to go into remission? Scientists from the Institute of Diabetes Research and Metabolic Diseases (IDM) at Helmholtz Munich and the Department of Diabetology, Endocrinology, and Nephrology at the University Hospital of Tübingen, conducted a post-hoc analysis on participants with prediabetes from the Prediabetes Lifestyle Intervention Study (PLIS) to investigate this question.
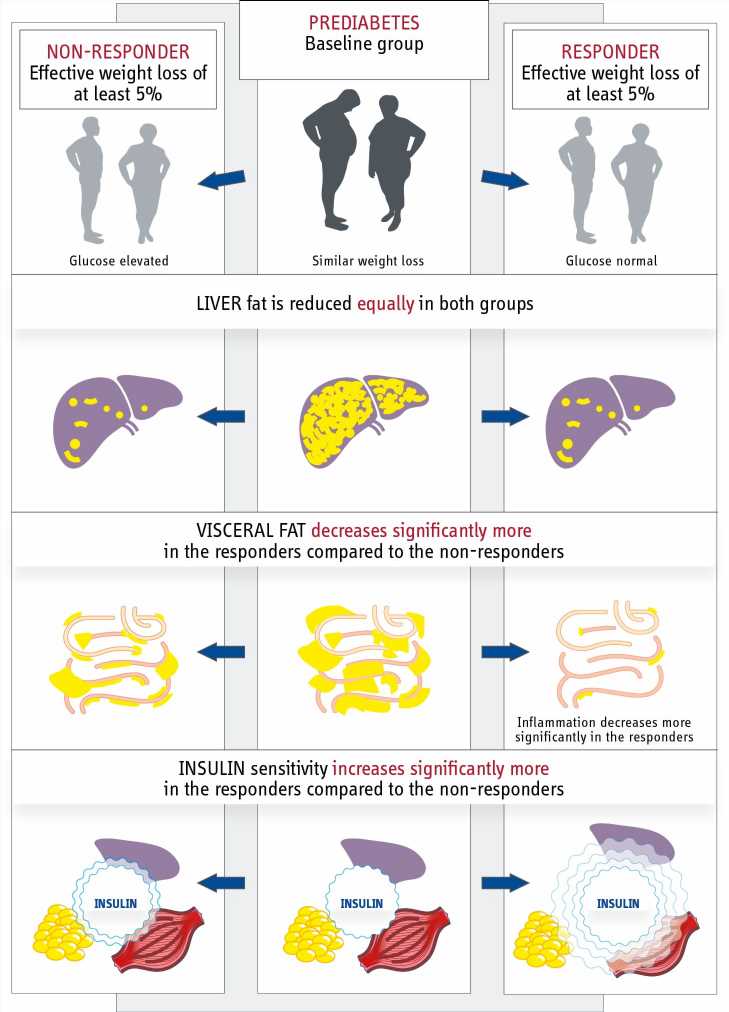
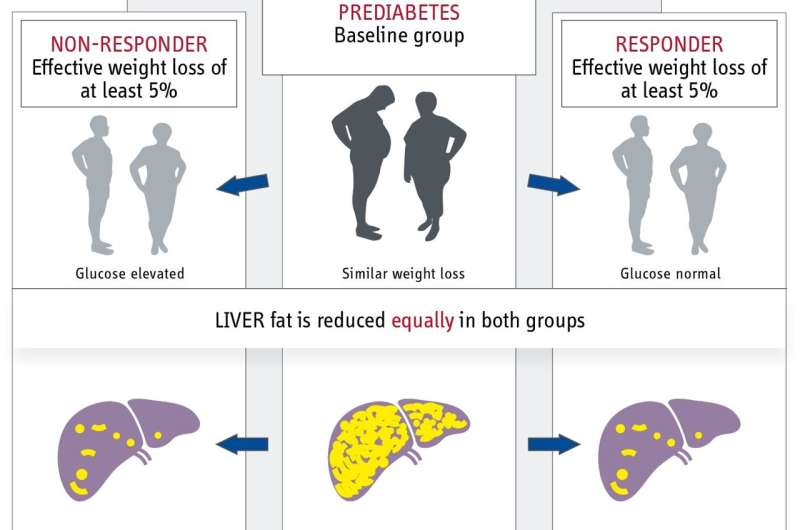
In this randomized-controlled multicenter study conducted by the DZD, 1,105 individuals with prediabetes underwent a lifestyle intervention involving a healthy diet and increased physical activity for a duration of one year. The researchers then assessed the 298 participants who had achieved a minimum weight loss of 5% as a result of the intervention.
Responders were the participants whose fasting blood glucose, 2-hour glucose, and HbA1c levels had normalized within twelve months, indicating that they had gone into remission. Non-responders were individuals who did not achieve remission despite losing weight and still had prediabetes.
Contrary to the researchers’ initial assumptions, it was not weight loss that distinguished those who went into remission from those who did not, as there was no difference in relative weight loss between responders and non-responders. However, individuals who achieved remission demonstrated a notable improvement in insulin sensitivity compared to non-responders. In essence, they were able to enhance their sensitivity to insulin, a hormone that lowers blood glucose levels, significantly more than those who did not respond.
Nonetheless, the quantity of insulin secreted remained unaltered in both groups. This difference is critical compared to type 2 diabetes remission, which depends primarily on enhanced insulin secretion.
Reducing abdominal fat mass may help reverse prediabetes
To determine the cause of increased insulin sensitivity in responders, the researchers conducted a comparative analysis of the two groups. The responders had lost more abdominal fat compared to non-responders despite losing the same amount of body weight. Visceral abdominal fat is located directly in the abdominal cavity and surrounds the intestines. Its impact on insulin sensitivity is partially attributed to an inflammatory response in adipose tissue.
Indeed, participants who went into remission also had fewer inflammatory proteins in their blood. “Since the responders showed a reduction in abdominal fat in particular, it will be important in the future to identify the factors that promote the loss of this fat depot,” says Arvid Sandforth, one of the two lead authors. Surprisingly, there were no differences between the two groups in the reduction of liver fat, which is also an important risk factor for the development of diabetes.
Participants who achieved remission showed a 73% reduced risk of developing type 2 diabetes even two years after the end of the lifestyle intervention. They also showed reduced markers of kidney damage and better status of their blood vessels.
Currently, treatment of prediabetes consists of weight reduction and lifestyle improvement to delay the onset of type 2 diabetes—but without glucose-based targets to guide the treatment process. The DZD’s new analysis fills this gap. “Based on the new data, remission should be the new therapeutic target in people with prediabetes. This has the potential to change treatment practice and minimize the complication rate for our patients,” says co-first author Prof. Dr. Reiner Jumpertz-von Schwartzenberg.
According to the study, remission in prediabetes can be considered to have occurred when fasting blood glucose falls below 100 mg/dl (5.6 mmol/l), 2-hour glucose below 140 mg/dl (7.8 mmol/l), and HbA1c below 5.7%. The likelihood of remission increases when body weight is reduced and waist circumference decreases by at least about 4 cm in women and about 7 cm in men. Researchers state that these criteria can now be used as biomarkers.
More information:
Arvid Sandforth et al, Mechanisms of weight loss-induced remission in people with prediabetes: a post-hoc analysis of the randomised, controlled, multicentre Prediabetes Lifestyle Intervention Study (PLIS), The Lancet Diabetes & Endocrinology (2023). DOI: 10.1016/S2213-8587(23)00235-8
Journal information:
The Lancet Diabetes & Endocrinology
Source: Read Full Article