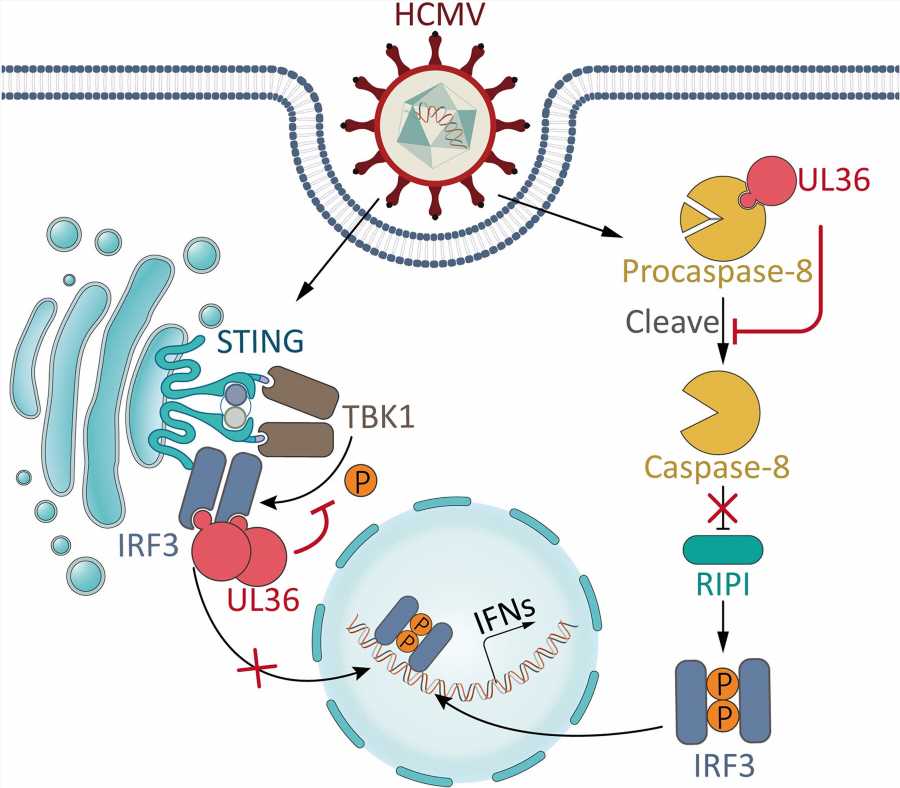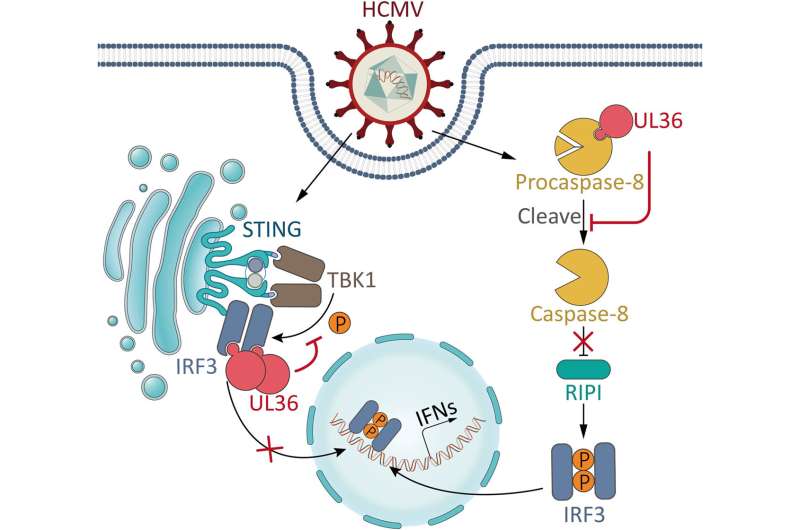
Human cytomegalovirus (HCMV) is a β-herpesvirus that infects most of the world’s population and poses a serious threat to human health. The constant exposure of HCMV to the innate immune system makes this virus evolve a rich arsenal of immune evasion strategies.
In addition to innate immunity, apoptosis also acts as an important antiviral mechanism by eliminating infected cells. Thus, inhibition of apoptosis is also of critical importance for HCMV. Among HCMV-encoded proteins, UL36 has long been identified as an inhibitor of extrinsic apoptotic pathway by directly binding and inhibiting caspase-8.
A previous study reported that caspase-8 can inhibit IRF3 activation. Therefore, although caspase-8/apoptotic inhibition by HCMV-encoded UL36 may provide a favorable cellular environment for HCMV infection, it would inevitably result in enhanced antiviral immune signaling that is detrimental for this virus. Such a “side effect” brings a dilemma of how to balance immune evasion and anti-apoptosis, the two pivotal requirements for infection by HCMV and other viruses encoding apoptotic inhibitors.
In a new study, a research group led by Prof. Zhou Xi from the Wuhan Institute of Virology of the Chinese Academy of Sciences (CAS) showed that in addition to inhibiting caspase-8/extrinsic apoptosis, HCMV UL36 antagonizes IRF3-dependent immune signaling by directly targeting IRF3. UL36 confers these two inhibitory functions simultaneously during HCMV infection.
The study was published in Science Advances, titled “Human cytomegalovirus UL36 inhibits IRF3-dependent immune signaling to counterbalance its immunoenhancement as apoptotic inhibitor.”
While the inhibition of caspase-8/extrinsic apoptosis by UL36 enhances immune signaling, the immunosuppressing activity of UL36 counterbalances this immune-enhancing “side effect” for viruses, and the dual inhibition by UL36 is of particular functional importance for the HCMV life cycle.
It is worth noting that a previous study by the same group uncovered that another HCMV protein, UL37x1, also confers dual inhibition on both apoptosis and innate immunity.
Unlike UL36, UL37x1 is an inhibitor of intrinsic apoptosis, while UL37x1 inhibits immunity by targeting TBK1. Thus, the same virus encodes two distinct proteins, UL36 and UL37x1, to inhibit extrinsic and intrinsic apoptosis, respectively, highlighting the critical value of establishing a sophisticated regulatory network to inhibit both extrinsic and intrinsic apoptotic pathways and their corresponding immunoenhancing effects, thereby ensuring a balanced, favorable cellular microenvironment for HCMV.
Given that most viruses (not limited to herpesviruses) use their own strategies to inhibit apoptosis, it would be intriguing to find out how these viruses achieve such a balance between immune evasion and anti-apoptosis.
More information:
Yujie Ren et al, Human cytomegalovirus UL36 inhibits IRF3-dependent immune signaling to counterbalance its immunoenhancement as apoptotic inhibitor, Science Advances (2023). DOI: 10.1126/sciadv.adi6586
Journal information:
Science Advances
Source: Read Full Article