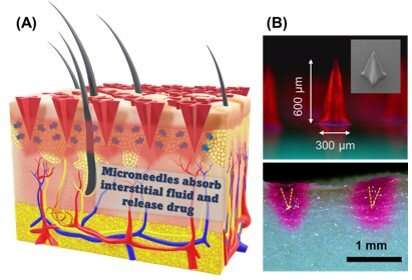
The painful feeling of receiving an injection through a hypodermic needle or with the unpleasant sensation of swallowing a large pill is a globally familiar sensation. But what if a revolutionary and gentler way of administering drugs was in the works? For over two decades, researchers have been investigating various types of microneedles as a minimally invasive method for transdermal drug delivery. Arrays of microneedles can be designed to be loaded with a drug or chemical, which they then release over time onto the blood stream after piercing slightly beyond the skin layers.
Microneedles offer several advantages compared to other types of drug delivery. First, they are painless and cause virtually no damage to the skin nor bleeding. Second, they can be self-administered. Third, unlike traditional needles, the disposal of microneedles is much easier as they don’t leave behind hazardous waste. Unfortunately, there are still a few challenges that need to be addressed before microneedles become the next big thing in healthcare. One is their fabrication cost, which generally involves expensive molds, materials, and machinery. Another issue is the aggregation and degradation of proteins when microneedles are pre-loaded with a protein-based medicine, as these molecules are quite sensitive to external conditions such as temperature, acidity and salt concentration.
In a recent study published in Biomacromolecules, two research teams from Japan and Thailand collaborated to address the main limitations of existing microneedles. On the one side, Prof. Kazuaki Matsumura from the Japan Advanced Institute of Science and Technology (JAIST) and Ph.D. student Harit Pitakjakpipop from JAIST-SIIT developed and applied a functional polymer that effectively suppresses protein aggregation. On the other side, Dr. Paisan Khanchaitit and his team at the National Science and Technology Development Agency (NANOTEC), Thailand, perfected a microneedle fabrication method suitable for the industrial scale based on photolithography. By combining these two efforts, the teams managed to produce microneedles patches with several attractive properties and potential scalability to clinical settings.
The microneedles themselves are made of a non-degradable, biocompatible hydrogel that also contains zwitterionic poly-sulfobetaine (poly-SPB). As reported in previous studies by the same authors, this polymer suppresses protein aggregation. Thus, the researchers incorporated it during the fabrication process and showed that the proteins pre-loaded in the microneedles were stable even when subjected to various external stresses.
Additionally, the scientists developed a straightforward and cost-effective way to fabricate microneedle arrays made from the abovementioned materials. They resorted to photolithography, a process in which a photomask is used to selectively block UV light from reaching a target surface to control chemical reactions locally, as Dr. Khanchaitit explains: “UV light passing through the photomask generates free radicals in the polymer resin during the fabrication process, resulting in photopolymerization and the subsequent formation of 3D microneedle structure patterns on a clear and flexible substrate.” This fabrication procedure requires inexpensive equipment only and takes about five minutes, yet produces four-point star-shaped microneedles with remarkable mechanical strength.
To test the performance of these microneedle arrays for drug delivery, the researchers loaded them with 50 microliters of drug solutions containing rhodamine B as a dye alongside lysozyme and insulin as example proteins. Through various experiments on porcine skin, the teams verified that their microneedle patches offered both high drug-loading capacity and high drug-release rate. Moreover, they confirmed that the microneedles could both load and preserve various water-soluble drugs and proteins simultaneously, eliminating the need for refrigeration.
Source: Read Full Article
